Amplia Therapeutics Limited (ASX: ATX), (“Amplia” or the “Company”), announces mature data from the ongoing ACCENT clinical trial in advanced pancreatic cancer in which the Company’s lead drug narmafotinib is combined with chemotherapy showing a median overall survival of 11.1 months, and five complete responses recorded to date.
HIGHLIGHTS
- Formal centralised and independent analysis of the clinical response data from the ACCENT trial has been undertaken and updated analysis provided to the Company
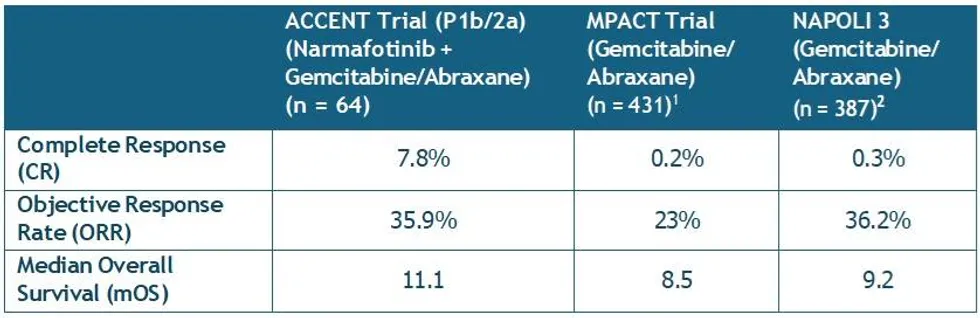
- The data confirm that five (5) patients have achieved a complete response (CR) in the Phase 1b/2a trial of narmafotinib combined with chemotherapy resulting in an unprecedented rate of CRs of 7.8% (5/C4)
- A median Overall Survival of 11.1 months has also been determined which is a 2 month improvement compared to chemotherapy alone, with no additional toxicity burden
- The Company has been chosen to present trial data at the prestigious annual meeting of the American Association of Cancer Research (AACR) being held in April 202C in San Diego, CA, USA
Expert central reading of the clinical data by a contracted independent laboratory has reclassified some of the response data, identifying an additional four (4) confirmed complete responses (CRs). This brings the total CR’s for all patients in the ACCENT trial receiving a 400 mg dose of narmafotinib to five (5), resulting in a CR rate of 7.8% (5/64) which is unprecedented in this indication. Notably, this does not include the pathological complete response (pCR) recorded in the ACCENT trial, announced in June 2025. A confirmed CR means that CT scans have confirmed the disappearance of measurable tumours and metastases for two months or more, without the appearance of new lesions.
An additional confirmed partial response (PR) has also been identified, resulting in an updated Objective Response Rate (ORR) of 35.9% (23/64) for all patients in both stages of the 1b/2a ACCENT trial on a 400 mg dose of narmafotinib. As of 15 March 2026, four (4) patients remain on study, with one patient approaching 24 months on trial.
Up until the independent analysis, all clinical response data reported to the market has been based on analysis by the clinical investigator at each trial site. The Company has always planned for an independent data analysis to occur toward the conclusion of the trial, and with the anticipated completion in Ǫ3 2026 this analysis was recently initiated. The expert and independent ‘central read’ laboratory has used the standardized and internationally recognized RECIST 1.1 criteria for measuring how a patient’s cancer responds to treatment.
Importantly, analysis of overall survival data (with a data cut-off of mid-March), indicates a median Overall Survival (mOS) of 11.1 months. This is an approximate two-month improvement when compared to clinical studies of the gemcitabine-Abraxane® chemotherapy alone, including the MPACT study1, which established this standard-of-care for advanced pancreatic cancer and against which ACCENT is benchmarked.
Combined, these data compare very favourably to published data for the gemcitabine-Abraxane chemotherapy alone from both the historical MPACT trial1 and the recent NAPOLI 3 trial2 (see table). Particularly noteworthy is that the mOS data from the ACCENT trial is identical to that obtained for the combination chemotherapy regimen NALIRIFOX in NAPOLI 3, and which resulted in its subsequent approval by the US FDA.
Narmafotinib continues to be well tolerated by patients with the adverse effect profile of the narmafotinib- chemotherapy combination similar to chemotherapy alone.
The Company has been selected to present its trial data, along with additional ACCENT data derived from further analysis of the independently read data, at the annual meeting of the American Association of Cancer Research (AACR) meeting to be held April 17-22 in San Diego, California.
Dr Chris Burns, CEO and Managing Director of Amplia, commented on the latest results: “These latest data from the ACCENT trial clearly demonstrate the significant clinical benefit of narmafotinib. The unprecedented 7.8% rate of CR’s in the first line setting provides new hope for patients with this very aggressive cancer and provides further strong support for the benefit that narmafotinib can bring when combined with other treatment modalities. We look forward to presenting a detailed analysis of the ACCENT trial at the forthcoming AACR conference.”
Click here for the full ASX Release
This article includes content from Amplia Therapeutics, licensed for the purpose of publishing on Investing News Australia. This article does not constitute financial product advice. It is your responsibility to perform proper due diligence before acting upon any information provided here. Please refer to our full disclaimer here.
Read the full article here
